The Araujo Laboratory: Lysosomal signaling complexes – Coordinating organelle function with cellular homeostasis
Lysosomes are membrane bound organelles that fulfill several functions, from protein degradation to the control of cellular metabolism. Due to their importance for overall proteostasis and signaling, cells have developed unique mechanisms to control lysosomal numbers, size, movement, degradative capacity, interaction with other organelles, and even lysophagy, the name given to the selective degradation of dysfunctional lysosomes. The importance of this organelle is highlighted by the variety of diseases known to be associated with lysosomal abnormalities, from monogenic lysosomal storage disorders to brain pathologies, metabolic disorders, cancer and aging.
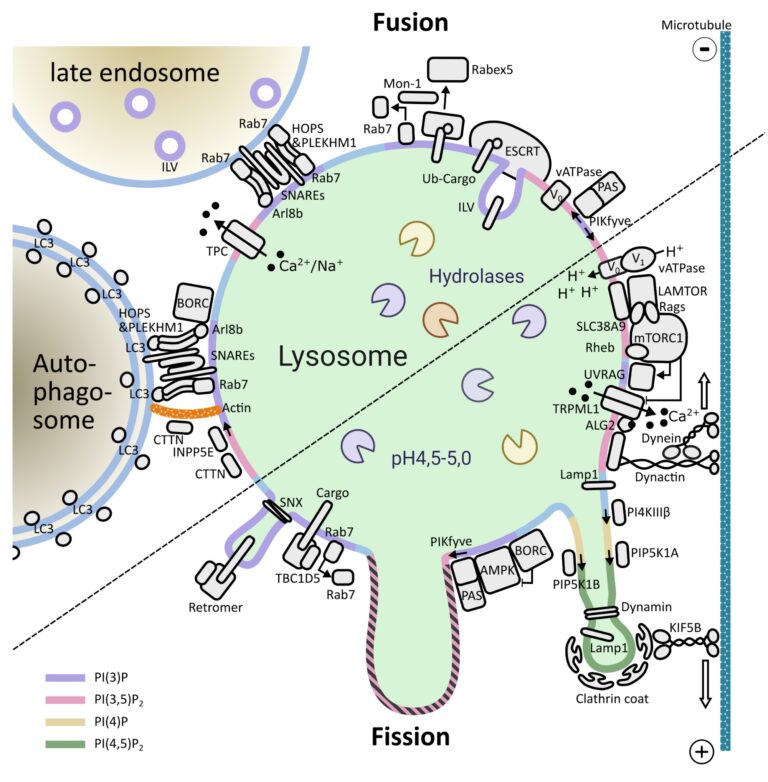
One of the key signaling complexes present in the lysosomal membrane is mTORC1, Mechanistic target of rapamycin complex 1. It regulates the balance between biosynthetic and catabolic processes influencing a plethora of cellular processes. Because mTORC1 activation triggers a fairly resource-intensive anabolic program, cells have evolved mechanisms to ensure that it only becomes active when sufficient resources are available. Over the last years, work from a number of laboratories have elucidated many aspects mTORC1’s regulation: from identifying the key players, to deciphering the molecular mechanisms involved and the three-dimensional structure of these assemblies.
Building on this vast pool of knowledge, we focus on the interplay between canonical mTORC1 signaling components and other lysosomal proteins/complexes.
Our objectives are three fold:
1) Decipher how these molecular machineries coordinate their activity and help control the organelle’s properties and function.
2) Determine the implication of these events for overall cellular homeostasis.
3) Understand how dysfunction of these processes underlies different disease conditions.
To address the above, we combine genome editing with cell biology techniques, biochemical and imaging approaches. We also actively collaborate with structural biology groups, to shed light on specific molecular mechanisms controlling our favorite proteins.
Publications
https://pubmed.ncbi.nlm.nih.gov/?term=de%20Araujo%20MEG&sort=date
https://scholar.google.com/citations?hl=en&user=5jH0XooAAAAJ&view_op=list_works&sortby=pubdate
Selected publications
- de Araujo MEG*, Amann S, Stasyk T, Schleiffer A, Rauch E, Flümann P, Singer I, Kremser L, Dostal V, Laopanupong T, Obojes N, Wallnöfer MH, Gradl FS, Kurzbauer R, Krebiehl C, Kofler S, Grishkovskaya I, Vogel GF, Hess MW, Sarg B, Clausen T, Haselbach D, Huber LA*. BORC assemblies integrate BLOC-1 subunits to diversify endosomal trafficking functions. Proc Natl Acad Sci U S A. 2026 Jan 27;123(4):e2515691123. doi: 10.1073/pnas.2515691123. Epub 2026 Jan 20. PMID: 41557793. * Shared corresponding author
- Cui Z, Napolitano G, de Araujo MEG,
Esposito A., Monfregola J, Huber LA, Ballabio A
and Hurley JH. Structure of the lysosomal mTORC1-TFEB-Rag-Ragulator mega complex. Nature 2023, Feb;614(7948):572-579. doi: 10.1038/s41586-022-05652-7. PMID: 36697823.
- Napolitano G, Di Malta C, Esposito A, de Araujo MEG, Pece S, Bertalot G, Matarese M, Benedetti V, Zampelli A, Stasyk T, Siciliano D, Venuta A, Cesana M, Vilardo C, Nusco E, Monfregola J, Calcagnì A, Di Fiore PP, Huber LA, Ballabio A. A substrate-specific mTORC1 pathway underlies Birt-Hogg-Dubé syndrome. Nature. 2020 Jul 1. doi: 10.1038/s41586-020-2444-0. PMID:32612235.
- de Araujo MEG, Liebscher G, Hess MW, Huber LA. Lysosomal size matters . Traffic. 2020 Jan;21(1):60-75. doi: 10.1111/tra.12714. Epub 2019 Dec 6. Review. PMID:31808235.
- de Araujo MEG, Naschberger A, Fürnrohr BG, Stasyk T, Dunzendorfer-Matt T, Lechner S, Welti S, Kremser L, Shivalingaiah G, Offterdinger M, Lindner HH, Huber LA, Scheffzek K. Crystal structure of the human lysosomal mTORC1 scaffold complex and its impact on signaling . Science. 2017 Sep 21. pii: eaao1583. doi: 10.1126/science.aao1583. PMID: 28935770
Collaborations
Gennaro Napolitano (Tigem, Naples, IT)
David Haselbach (IMP, Vienna, AUT)
Kathrin Thedieck (Institute of Biochemistry,University of Innsbruck, AUT)
Hesso Farhan (Institute of Pathophysiology, Medical University of Innsbruck, AUT)
Funding
FFG Eurostars nr 8706 2026 AUTOBIORES
FWF FG 2005/61357 2023- Organelle proteostasis in cellular quiescence and growth subproject5)
TWF GZ F16676/5 2019 – A potential new mechanism for selective modulation of mTORC1
Team and Contact

The Araujo team, January 2026 from left Lisa, Mariana, Thanida, Flora

Priv. Dozentin Mariana Eca Guimaraes de Araujo, PhD
Email:mariana.araujo@i-med.ac.at
Phone: +43 512 9003 70174
Google scholar: https://scholar.google.com/citations?hl=en&user=5jH0XooAAAAJ&view_op=list_works&sortby=pubdate
Name
Mariana Eca Guimaraes de Araujo
Thanida Laopanupong
Flora Gradl
Lisa Egger
Position
Group leader
PhD student
PhD student
Master student – Molmed